- Preparations for bees
- Feeds and additives for bees
- Containers for honey and buckets
- Wax and wax melters
- Beehives
- BeeBox
- Polystyrene foam hive
- Honey Tank and buckets
- Souvenirs
- Seeds
- Equipment for beekeeping
- Uncapping cells
- Frames and accessories
- Queen bees and equipment
- Processing honey
- Clothes of the beekeeper
- Honey extractors
- Preparations for bees
- Feeds and additives for bees
- Containers for honey and buckets
- Wax and wax melters
- Beehives
- BeeBox
- Polystyrene foam hive
- Honey Tank and buckets
- Souvenirs
- Seeds
- Equipment for beekeeping
- Uncapping cells
- Frames and accessories
- Queen bees and equipment
- Processing honey
- Clothes of the beekeeper
- Honey extractors
- Catalog
- Preparations for bees
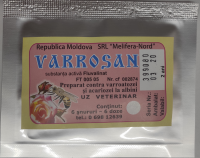
- Fluvalidez
Catalog
- Preparations for bees
- Feeds and additives for bees
- Containers for honey and buckets
- Wax and wax melters
- Beehives
- BeeBox
- Polystyrene foam hive
- Honey Tank and buckets
- Souvenirs
- Seeds
- Equipment for beekeeping
- Uncapping cells
- Frames and accessories
- Queen bees and equipment
- Processing honey
- Clothes of the beekeeper
- Honey extractors
Composition and form of release:
Dosage form:
Veterinary plate
Fluvalidez in 1 plate contains as active ingredients: fluvalinate - 80 mg, lavender essential oil - 60 mg, thyme essential oil - 60 mg, rosemary essential oil - 80 mg, and as auxiliary substances – ethyl cellulose and shelled veneer.
In appearance, the drug is a wooden plate from light beige to brown in color with the smell of essential oils.
Packaging:
The drug Fluvalidez is produced packaged in 10 plates in hermetically sealed bags made of metallized film. Each consumer package is provided with instructions for use.
Expiration date:
The shelf life of the medicinal product, subject to storage conditions in the manufacturer's closed packaging, is 3 years from the date of production.
It is prohibited to use the drug after the expiration date.
After opening the package, the medicinal product is not subject to storage.
Storage conditions:
The drug is stored in the manufacturer's closed packaging, in a place protected from direct sunlight, separately from food and feed at a temperature from 0 ° C to 25 ° C.
The drug should be stored in places inaccessible to children.
Doses and method of application:
The drug Fluvalidez is used for the treatment and prevention of varroatosis and acarapidosis of honey bees.
Fluvalidez is used in the spring and autumn periods. The drug is placed in hives at the rate of 2 plates for 10-12 nest frames. For small families (up to 6 frames), layering and nuclei, 1 plate is enough, which is placed in the center of the beehive. In a weak family, one plate is suspended between 3 and 4 frames, in a strong one - one plate between 3 and 4 frames, the second plate - between 7 and 8 frames. The plates are left in beehives for a period of 3 to 30 days, depending on the amount of printed brood, after which they are removed.
Safety precautions:
The use of the medicinal product is prohibited less than 14 days before the start of the main honey collection in order to avoid the ingress of the components of the medicinal product into commercial honey.
When working with a medicinal product, it is necessary to observe the general rules of personal hygiene and safety provided for when working with medicinal products for veterinary use. Empty packaging from under the drug is prohibited to be used for household purposes, it is subject to disposal with household waste.
In case of accidental contact of the drug with the skin or mucous membranes of the eyes, they must be rinsed with plenty of water. People with hypersensitivity to the components of the drug should avoid direct contact with it. In case of allergic reactions or accidental ingestion of the components of the drug into the human body, you should immediately contact a medical institution (have the instructions for use or label with you).
The drug is forbidden to be used by bees during the main honey harvest, there are no age restrictions.